Successful co-creation: How can an AI specialist and a pharmaceutical company drive innovation together?
Success factors for cooperation using the example of fully automated container inspection
A fully automated container control station has long been an idea for process optimization at the Freiburg production site – now the "Artificial Intelligence Container Check" is within reach: After the successful completion of the feasibility study, the project team, which is made up of employees from the Healthcare Hub and the specialist department, is working on the proof of concept. The aim of the project is to replace the visual inspection of cleaned bulk and bulk containers with a fully automated inspection. To this end, Pfizer is cooperating with the AI experts at Data Spree. "Through this collaboration, we are not only implementing advanced AI solutions, but also a culture of continuous innovation that is setting new standards for automated inspection processes," said Leonard Brix, founder of Data Spree.
How does such a cooperation project work? And what are the success factors of a cooperation between an AI company and an established pharmaceutical company?
Process optimisation through digital technologies is on the agenda at the Pfizer plant in Freiburg, one of the world's most modern production facilities for the pharmaceutical industry. The Pfizer Healthcare Hub supports the specialist departments in this process – this is also the case with the Artificial Intelligence Container Check.
The task
At the Pfizer production plant in Freiburg, Germany, four different types of containers are used for powders, granules and substrates, which must be cleaned regularly. Since compliance with the Good Manufacturing Practice (GMP) guidelines and the high cleanliness requirements has the highest priority, highly effective washing systems are used. In addition, the cleaning result is checked by pharmaceutical technicians – this step is now to be replaced by fully automated control with the help of artificial intelligence.
Success factor 1: Courage to take (calculated) risks
"We knew that the task could not be solved with the help of simple sensors and cameras. At the beginning of the project, it was still unclear whether we would be able to get to grips with the complex challenges at all," says Marko Petrovic, project coordinator at the Healthcare Hub Freiburg. Nevertheless, the project team decided to invest time and money in order to examine whether an Artificial Intelligence Container Check could be implemented at the Freiburg production site.
Success factor 2: Flexibility for the framework conditions at Pfizer
For the implementation, the project team was looking for suitable cooperation partners. The most important criterion in the selection process is the willingness to flexibly adapt to the given conditions. In addition, GMP experience is an advantage, according to Dr. Hans Zebner, Project Engineer Innovation at the Healthcare Hub Freiburg.
Thanks to the good networking of the Pfizer Healthcare Hub Freiburg within various innovation ecosystems, several potential machine vision companies could be identified. The Pfizer project team chose Data Spree, a Berlin-based company with expertise in machine vision, AI and industrial automation. "The Data Spree team already had GMP experience and was looking directly for solutions to the challenges on site," says Dr. Hans Zebner. Parallel to the onboarding of the cooperation partner, the project team defined clear requirements, milestones and goals. These were translated into logical project steps, which then lead to the decision for or against the rollout at the Freiburg production site.
Feasible? Achievable? Profitable?
Project steps of the Artificial Intelligence Container Check
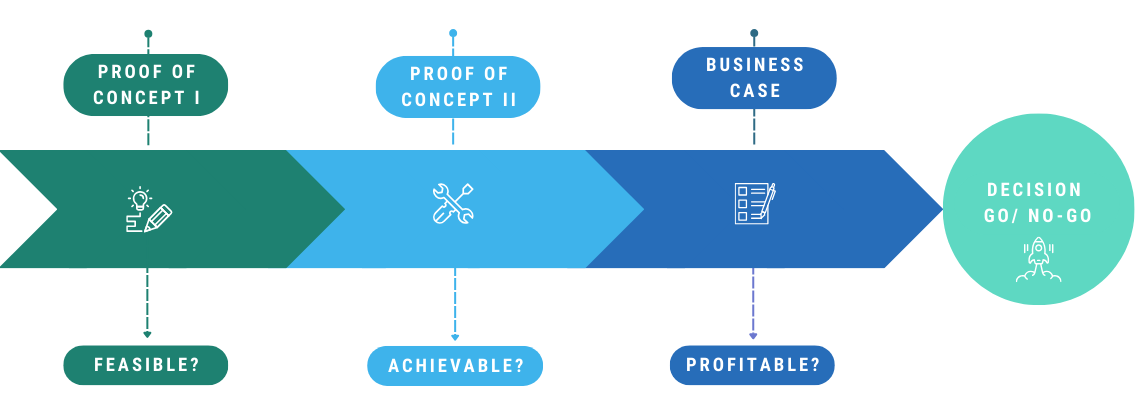
In the first part of the proof of concept, a feasibility study was carried out to show that simulated contaminants can be illuminated and photographed on the most technically demanding geometry of the NB granulate container in such a way that they can be easily identified.
Challenges of Proof of Concept I: Complex geometries and surfaces
The geometries of the four types of bulk and granulate containers used at the Freiburg production plant are complex: some of them have only very small openings. Different heights as well as irregularities of the surface structures due to dents, abrasive marks or weld seams in the interior make automated detection difficult. In addition, the container interiors are not rotationally symmetrical, but only mirror-symmetrical. For this purpose, the experts from Data Spree used an optical inspection system with a camera with a liquid lens lens and an illumination system with various light sources.
The interiors of the containers were photographed the interiors first in a clean condition, then with simulated contaminants. Adhesive surfaces of various sizes up to almost invisible adhesive threads served as contaminants.

Feasible!
The result: The simulated deviations were optimally illuminated, photographed and detected: dust grains, adhesive dots and adhesive threads below 0.5 mm could be identified. The image resolution of the camera exceeded the capabilities of the human eye. The feasibility study was thus considered to have been successfully completed.
"We at Data Spree are delighted with the good cooperation with Pfizer and the great commitment of all those involved. This project is a testament to how well it works to combine cutting-edge technology with industry knowledge to ensure the highest standards of quality and efficiency," said Leonard Brix.
Achievable?
The next step is to provide the proof of concept II for the AI tool in the interior and exterior of the container: The artificial intelligence is trained on defect characteristics with the help of real contaminants and must show that it is able to identify them.
Profitable?
Once the proof of concept has been provided, the specialist department will create a detailed project plan and calculate the business case. On this basis, the decision for or against the rollout at the Freiburg production plant is then made. "We would be delighted to realize this project at the Freiburg site. Not only because of the associated increase in productivity and the savings potential, but also because fully automated container inspection in the pharmaceutical sector is a real innovation," says Rainer Gut, responsible Subject Matter Expert.
Co-creation as a success factor
Willingness to take risks on the one hand, flexibility on the other – these are the decisive factors in the Artificial Intelligence Container Check in order to exploit the potential of innovative technologies in industrial applications. In addition, clearly defined requirements for the task, goal and process steps are a necessary basis for calculation and work for both sides. Co-creation as a model in which the partners involved contribute their respective strengths thus becomes a success factor for the digital transformation of production processes – we at the Pfizer Healthcare Hub are happy to support you!